|
4/23/2024 0 Comments Atomic theory timeline project This principal described the idea that as one gets closer to level of atoms and below the rules of motion start to warp. He created a principal known as the “uncertainty principal”. Heisenberg made a large contribution to the field of atomic theory with his ideas about the nature of subatomic particles in motion.His worked proved that the mass properties of atoms are not absolute and this would change the way in which elements needed to be studied from then on. He called these different forms of elements isotopes and found that they had everything in common with their regular counterparts except for an increased number of neutrons. Frederick theorized and proved that two atoms of the same element could have different masses. Frederick Soddy had a substantial influence on the modern atomic theory.This model would be used to make studying the atom easier and give a better understanding of its operation. This model involves a core which is the nucleus with orbits around the core of the electrons. He created a model of the atom which is the most well known and most accurate in history. Neils Bohr made huge contributions to the atomic theory.This information would prove valuable as more research on the nature of subatomic particles are conducted. He was able to use preexisting mathematics in order to fang the mass of the electron. Millikan was one of the pioneers of atomic theory.He called this particle the nucleus and it would serve as one of primary elements of future atomic models. From this observation Rutherford deduced that there must be a very small and dense particle in the center of the atom which is able to deflect the light. The laser appeared to bounce off the surface of the foil. In his experiments, Rutherford took a piece of gold and hammered it out until it was transparent and then shined a laser on it. Rutherford was one of the major influences in the modern atomic theory in that he discovered one of its key elements.This was the model that would for a while supersede the billiard ball model until refined and replaced. This model describes an atom as a glob of protons and neutrons in which the electrons are suspended. This structure of atom is known as the “plum pudding” model. He also created a new model for the structure of the atom. He found that all neutral partials of the same atom have the same number of negative particles. Thompson is widely recognized for having a large influence on the ideas of the atomic theory. He found that different types of elements release energy and decay into other elements. He discovered radiation and the three different classifications of nuclear radiation. 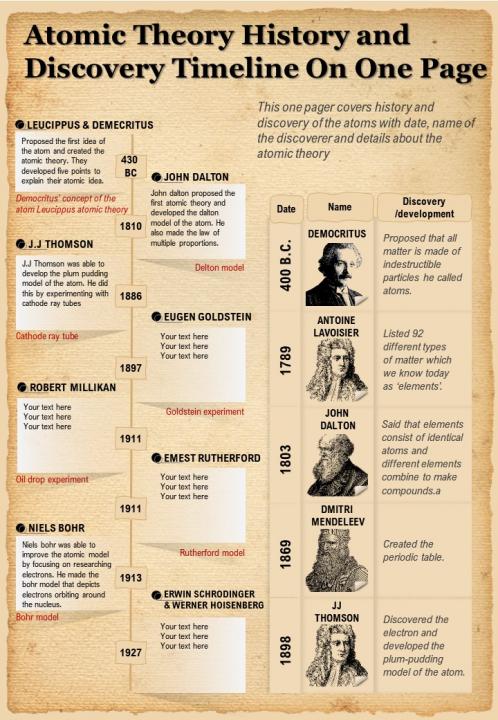 
He observed that certain elements give off an unseen energy which he called at the time Becquerel energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
